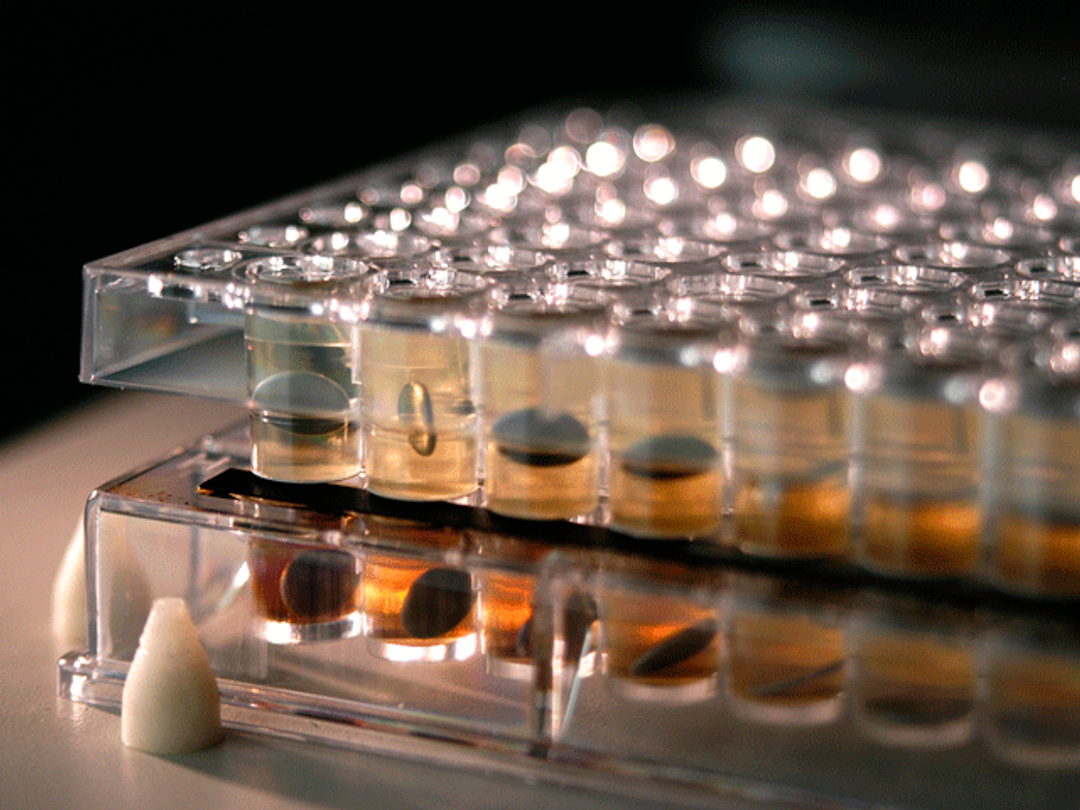
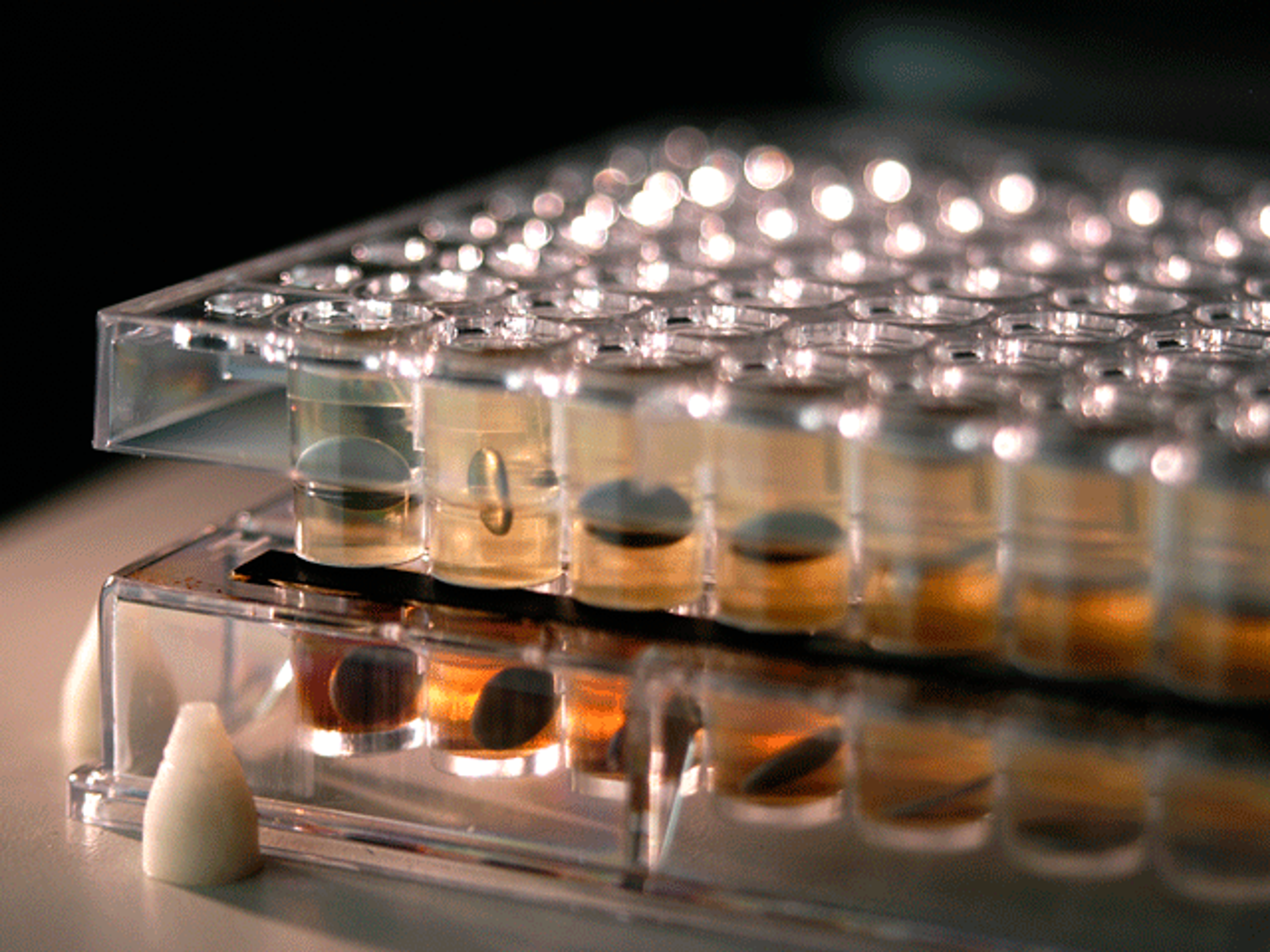
Parallel artificial membrane permeability assay (PAMPA) kits
Pion’s PAMPA high-throughput permeability assay uses a range of membranes to mimic physiologic conditions. It allows you to evaluate the passive permeability of API’s as well as the absorption potential of simple or complex formulations using robust models and protocols in a 96-well format.

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
Benefits:
- Evaluate the passive permeability of API's as well as the absorption potential of simple or complex formulations.
- Classification of API’s can be performed on BBB lipid for blood-brain barrier permeability in the CNS classification system
- Permeability classification of API’s can be performed on GIT lipid to support the Biopharmaceutical Classification System (BCS) ranking
Bio-Mimetic Artificial Membrane Permeation Through:
Gastro Intestinal Tract (GIT)
GIT PAMPA vs. Human Jejunal Permeability
The GIT PAMPA (Double-Sink™) model data was initially developed against eight compounds evaluated at the University of Uppsala with human subjects. Since that time, additional data have been added, providing increased confidence in the model.
Blood Brain Barrier (BBB)
BBB PAMPA vs. Rodent Brain Perfusion
Nearly 40 CNS compounds were used for building the BBB PAMPA model.
The developed model can predict rodent brain perfusion uptake rate better than most more expensive cell-based assays in the market.
Advancing oral drug development with in vitro dissolution and absorption tools
June 9, 2025 16:00 BST / 17:00 CEST / 11:00 EDT / 08:00 PDT
Effective absorption and therapeutic performance are essential to the success of oral small molecule drugs. In order to achieve this, understanding key biopharmaceutical properties such as solubility, dissolution, and membrane permeability are crucial for optimization as these factors can influence bioavailability and overall clinical efficacy.
Join Karl Box, Chief Scientific Officer at Pion Inc, as he explores innovative in vitro techniques that can help characterize key biopharmaceutical properties — enabling better decision-making from early discovery through to formulation optimization.
Designed for scientists and formulation teams, this exclusive webinar will highlight practical approaches to evaluating oral drug performance using dynamic dissolution and absorption models. Whether you are working on preclinical candidates or refining final dosage forms, gain insight into how real-time in vitro testing can be used to identify formulation risks early, troubleshoot challenges, and support a more efficient development process, to enhance the predictability and success of oral drug development.
Key learning objectives
- Understand how to apply small-scale and biorelevant dissolution testing to support formulation strategy.
- Explore tools and workflows for assessing supersaturation/precipitation and excipient selection.
- Gain insights into how in vitro data can guide risk assessment and improve developability profiling.
- Learn how to predict oral absorption (Fa%) and assign the BCS Class of your compound.
Who should attend?
- Pharmaceutical scientists involved in drug development and formulation.
- Preclinical development teams looking to improve biopharmaceutic assessments.
- Anyone interested in enhancing their understanding of oral absorption and developability assessment using in vitro methods.
- R&D professionals interested in in vivo predictive tools.
Certificate of attendance
If you attend the live webinar, you will automatically receive a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
If you view the on-demand webinar, you can request a certificate of attendance by emailing editor@selectscience.net.