Invitrogen™ mMESSAGE mMACHINE™ T7 mRNA Kit with CleanCap™ Reagent AG
Simplify synthesis and scale-up of potent mRNA with third-generation CleanCap®—proven technology for enhanced in vivo translatability.

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
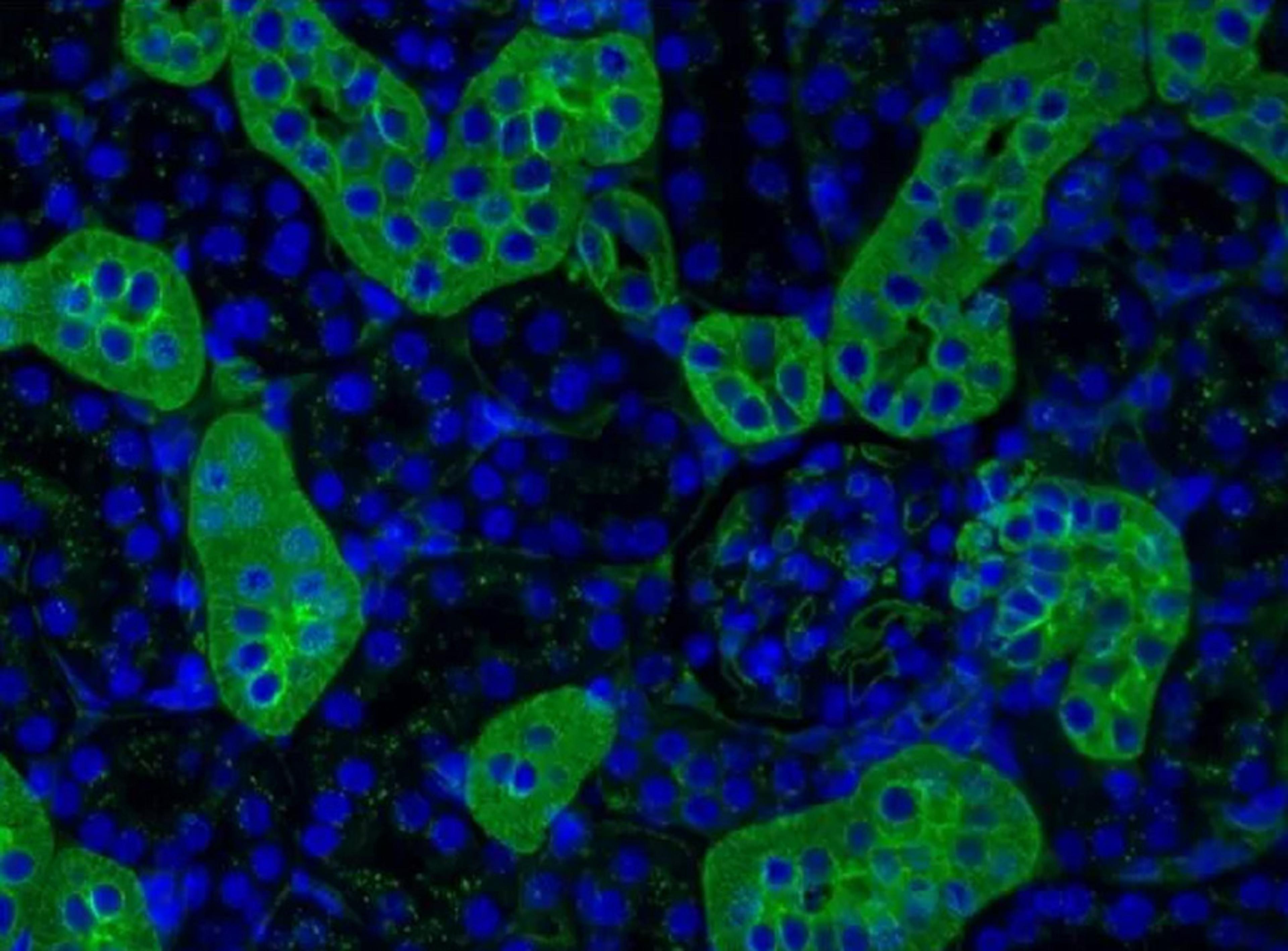
The mMESSAGE mMACHINE T7 mRNA Kit with CleanCap Reagent AG contains all the reagents and buffers necessary for in vitro synthesis of large amounts of mRNA with the naturally occurring Cap 1 structure for increased translatability in vivo compared to legacy capping methods such as the use of anti-reverse capping analog (ARCA). Utilization of the CleanCap Reagent AG ensures superior results relative to older capping technologies and greater protein production. Each standard 20-μL reaction yields a minimum of 100 μg of Cap 1-containing mRNA, while the scaled-up protocol yields up to 1 mg of mRNA. This kit is suitable for full- or partially-modified-nucleotide substitution to generate modified mRNA.
Key features:
- Contains proven CleanCap capping technology
- Optimized to achieve high mRNA yields (>5 mg/mL)
- Produces mRNA that is over 95% capped
- Optimized for production of mRNA from 0.9–10 kb templates
- Minimum yields of 100 μg per reaction, up to 150 μg per reaction (see figure below)
- Allows use of modified nucleotides, full- or partial-modified substitution
- Includes linearized 0.9 kb GFP control template, yields ≥100 μg mRNA
- Made up of high quality TheraPure reagents to enable transition from RUO to GMP
Invitrogen in vitro transcription kits - Flexible solutions to synthesize potent mRNA
With over 50,000 citations, the proven suite of Invitrogen™ in vitro transcription (IVT) kits are recognized as the most reliable and highest-performing reagents in the industry. In this application note, Thermo Fisher Scientific provides a guide to help you choose the kit that aligns with your experimental goals.
Generating high-quality mRNA-LNP complexes for in vivo delivery: Strategies and methods
Thursday, May 29, 2025 - 9:30 IST | 12:00 SGT | 13:00 KST, JST | 14:00 AEST
The production of high-quality mRNA for therapeutic applications presents several challenges, including yield limitation, low capping efficiency, stability concerns, and immunogenicity issues. In fact, crude mRNA preparations can trigger transient inflammatory responses, complicating their use in clinical settings.
In this expert webinar, we will discuss the latest advancements in mRNA synthesis, purification, and delivery technologies that are helping to overcome these barriers. This session will provide a practical walkthrough of using the mMESSAGE mMACHINE T7 mRNA Kit with CleanCap Reagent AG to achieve efficient co-transcriptional capping and generate high-yield, high-purity mRNA with minimal immunogenic contaminants.
You’ll also discover how Vivofectamine delivery solutions are utilized to encapsulate mRNA into lipid nanoparticles (LNPs) for stable and targeted in vivo delivery. We’ll highlight methods to assess the performance, expression, and immunogenic profile of these mRNA-LNP complexes through in vitro and in vivo assays, including luciferase reporter expression in mouse models.
Key learning objectives:
- Learn key steps in mRNA synthesis and purification
- Discover basic concepts and components in the formulation of LNPs
- Gain practical insights and protocols to enhance the efficacy and safety of mRNA-based treatments
Who should attend?
Researchers and professionals involved in mRNA therapeutics or applications.
Certificate of attendance
All webinar participants can request a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
If you view the on-demand webinar, you can request a certificate of attendance by emailing editor@selectscience.net.