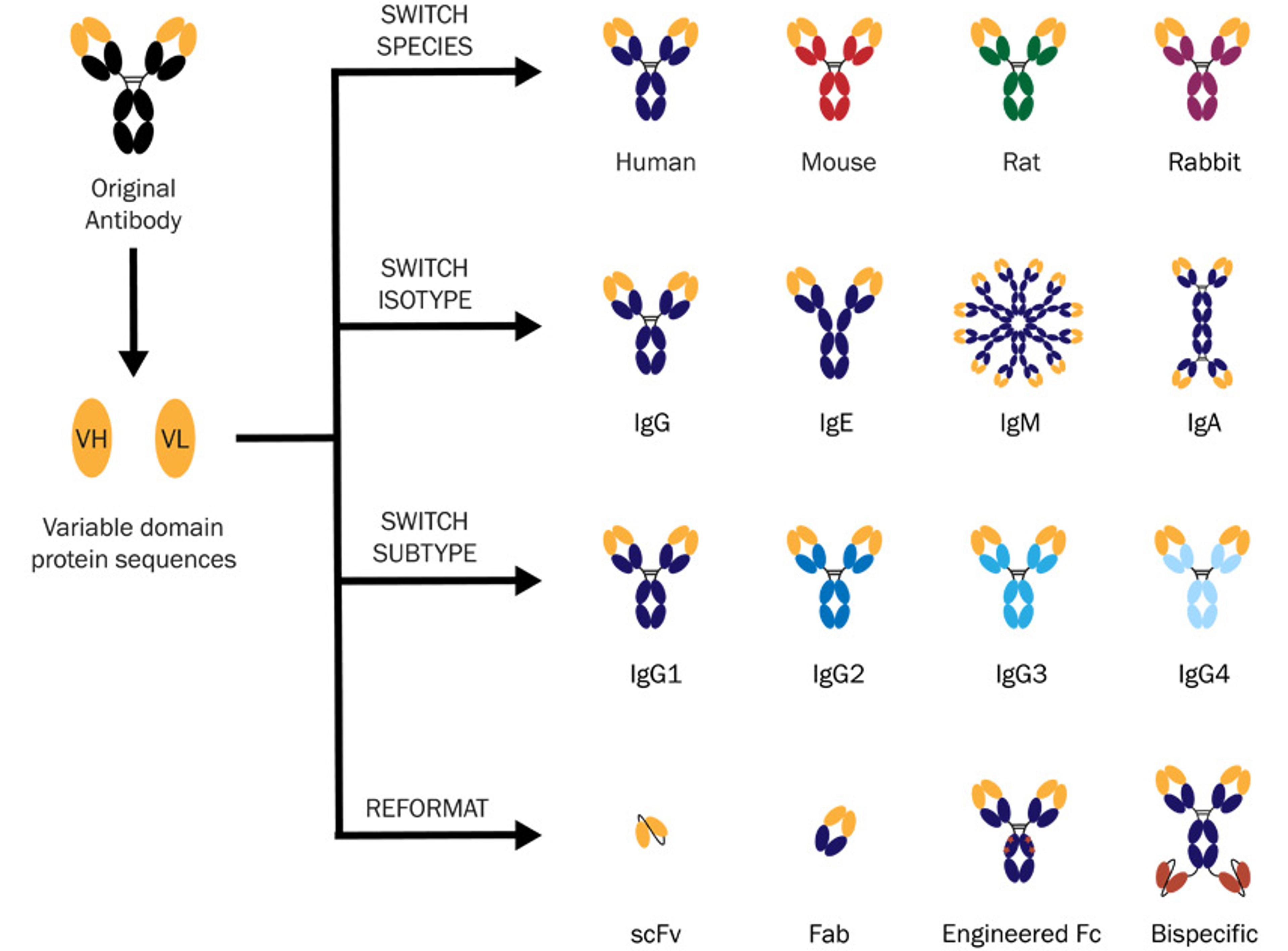
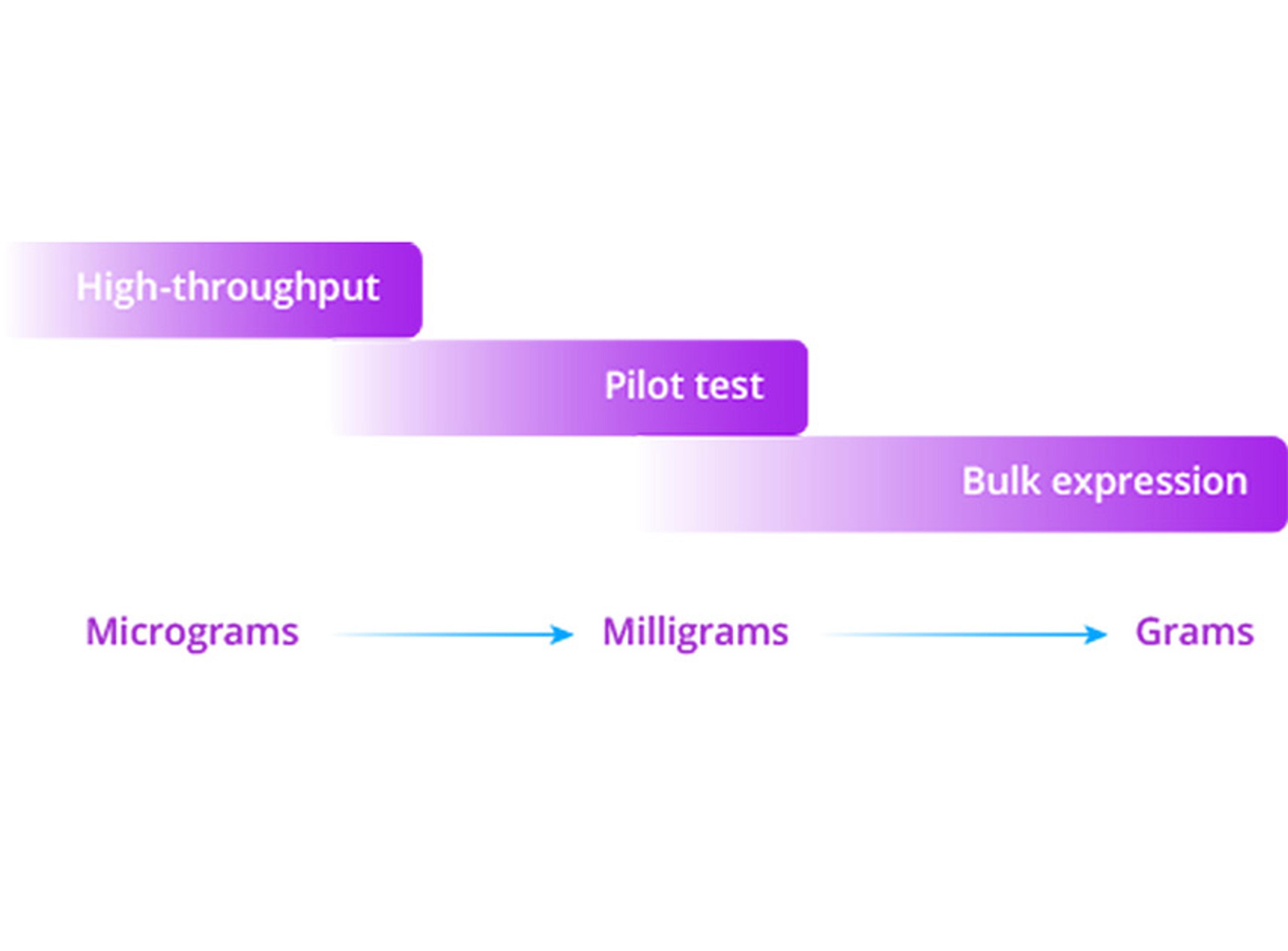
Discovery Services
iPSC drug discovery services from Ncardia

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
From hit finding to lead optimization, having a robust and human-relevant assay puts you on an ideal position to make data-driven decisions and mitigate risks
Beyond binding – iPSC-based functional safety screening for oncology modalities
Thursday, January 22 at 15:00 GMT | 16:00 CET | 10:00 EST | 07:00 PST
Biologics and cell-based therapeutics are expanding rapidly across oncology, yet early detection of safety liabilities remains a critical challenge. Off-target interactions and low-level antigen expression in healthy tissues can lead to severe clinical setbacks.
Recent FDA guidance emphasizes the need for human-relevant in vitro systems to assess functional toxicities before entering clinical trials. Induced pluripotent stem cell (iPSC)-derived models provide a reproducible, scalable solution for multi-tissue safety evaluation, including cardiac risk assessment.
In this webinar, industry experts will explore current challenges in oncology safety testing and the evolving regulatory landscape. They will introduce a novel iPSC-derived multi-tissue functional safety screening platform and demonstrate how comprehensive kinetic cardiac profiling can mitigate risk prior to clinical development, supported by case studies on antibody-drug conjugates (ADCs) and kinase inhibitors.
Key learning objectives
- Understand the current challenges and regulatory expectations for in vitro safety testing in oncology drug development.
- Learn how iPSC-derived multi-tissue models overcome limitations of traditional systems and improve translational relevance.
- Gain insights into integrating multi-tissue safety screening into drug development workflows and support regulatory alignment.
- Discover how early cardiac safety profiling can guide program decision and inform risk mitigation.
Who should attend?
- Big pharma: in vitro translational, pharmacology, toxicology and safety teams
- Biotechnology companies: in vitro translational, pharmacology, toxicology and safety teams
- Regulatory agencies employees: EMA and FDA
- Non-profit research organizations/foundations in oncology
- Oncology advocatory groups
Certificate of attendance
If you attend the live webinar, you will automatically receive a certificate of attendance, including a learning outcomes summary, for continuing education purposes.
If you view the on-demand webinar, you can request a certificate of attendance by emailing editor@selectscience.net.