Clinical Genomics Workspace
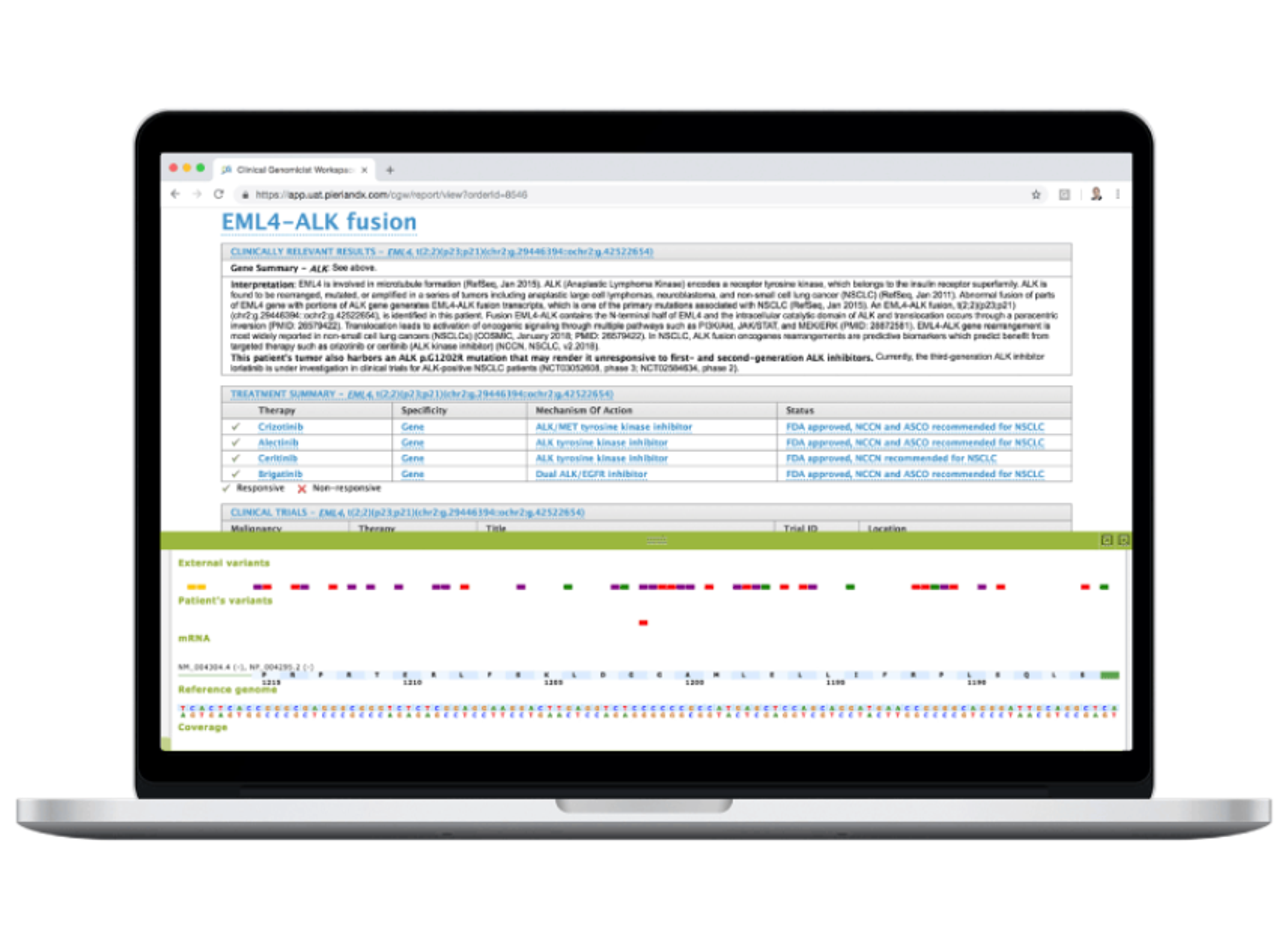
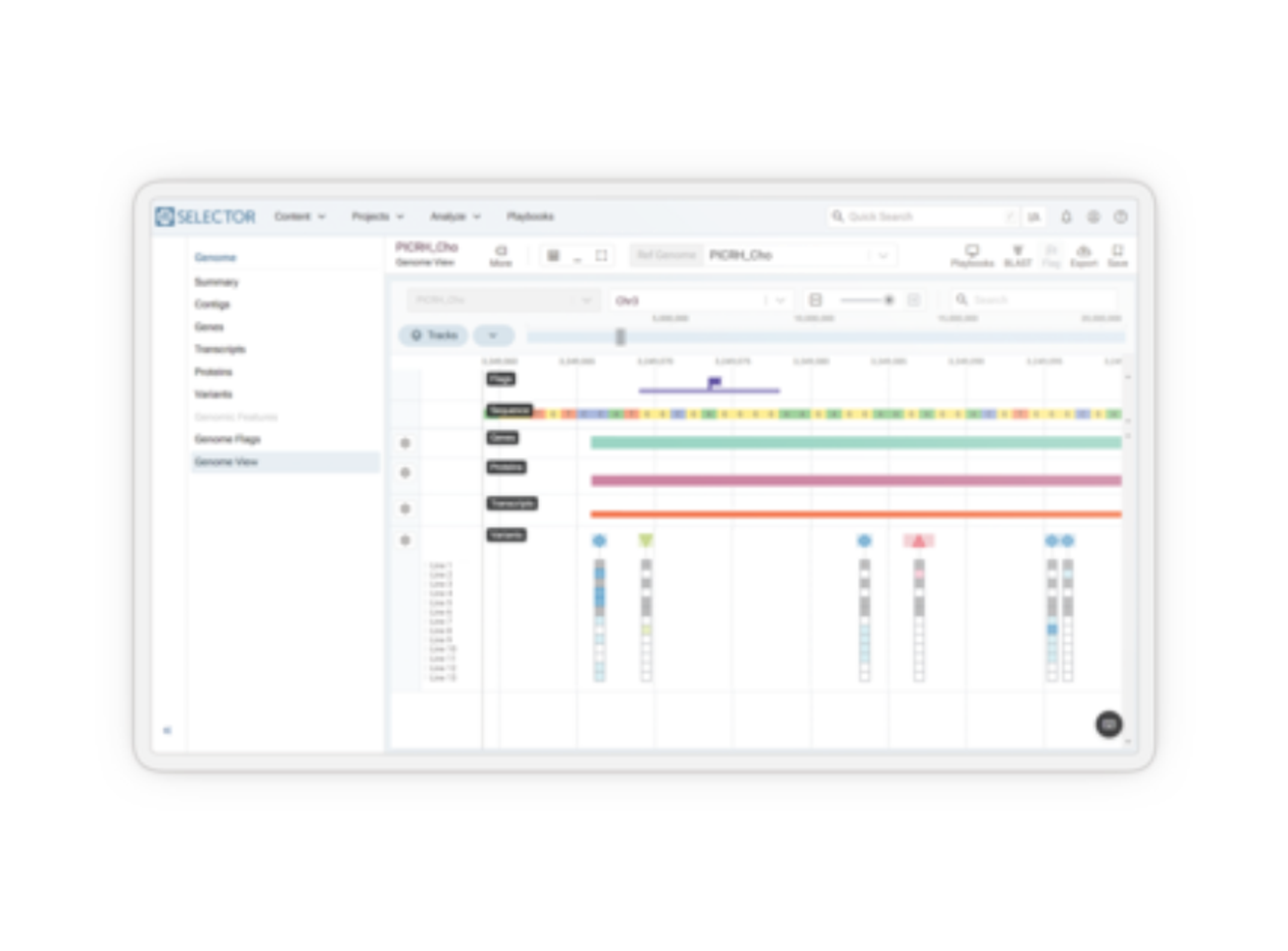
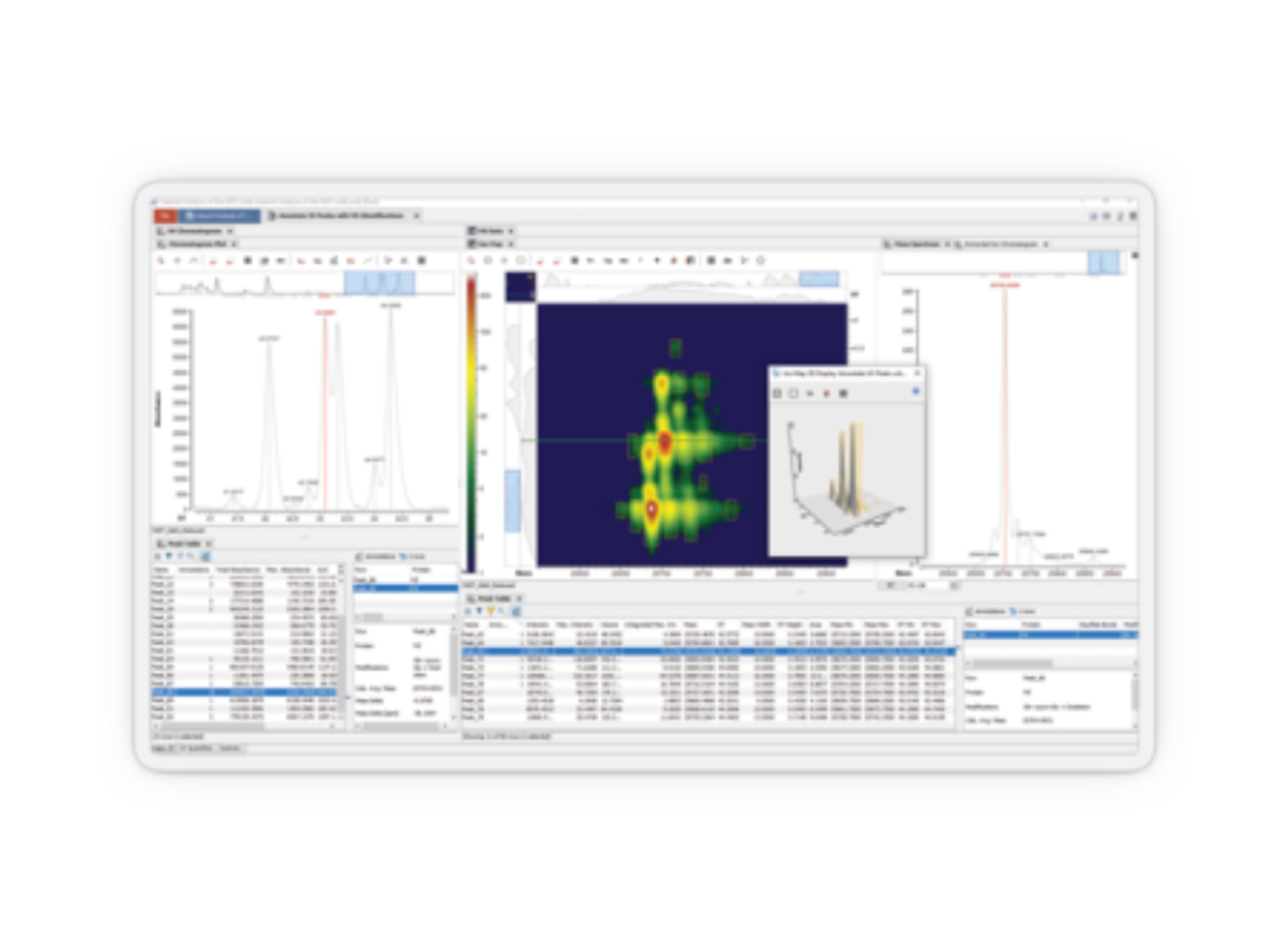
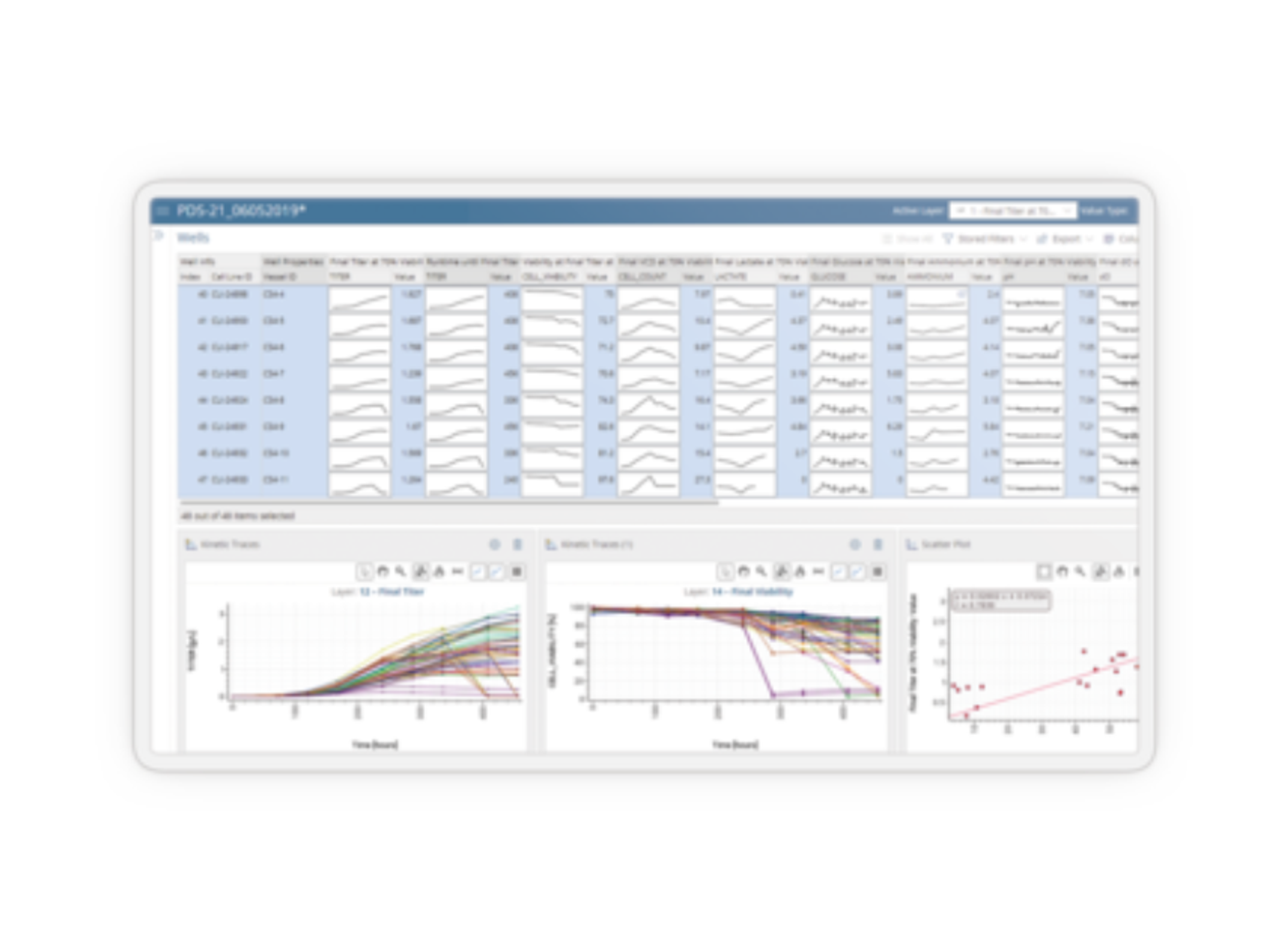
Disease, assay, instrument, and site-agnostic. Our end-to-end analysis and reporting solution streamlines your workflow from run and case level QA/QC review to rapid interpretation of NGS testing results, therapy selection, and clinical trial matching, so you can deliver the most precise medical management at every step of your patient’s journey.

The supplier does not provide quotations for this product through SelectScience. You can search for similar products in our Product Directory.
Speed up your NGS workflow without compromising on quality. Make actionable reports that elevate patient care. The Clinical Genomics Workspace (CGW) was purpose-built for physicians and clinical laboratory professionals in oncology and inherited disease to break down barriers to establishing or expanding their NGS testing program. From physician-owned laboratories to large hospitals to reference labs, CGW enables the success of molecular diagnostics experts around the globe.
Brochures
Overcome bottlenecks in NGS panel reporting
Next-generation sequencing (NGS) panel reporting presents challenges in efficiently identifying, reviewing, and reporting clinically relevant variants. With so many variants to assess, how can you ensure accuracy while optimizing turnaround time?
Inside this guide, you'll learn how to:
- Identify concise, accurate clinical content for reporting
- Quickly review large variant sets for quality and clinical relevance
- Build upon past decisions for faster, more consistent reporting
Find a solution that empowers you to:
- Customize quality and clinical evidence workflows
- Set preferences and customizations at the panel level
- Streamline filtering to focus on the most relevant variants
- Leverage a smarter knowledgebase for automated interpretations
- Facilitate VUS triage with variant pathogenicity insights
Develop and deliver your IVD with confidence
Bringing an in vitro diagnostic (IVD) solution to market requires precision, efficiency, and regulatory confidence. Discover how Velsera's advanced informatics platform and Clinical Genomics Workspace (CGW) provide a seamless, end-to-end solution for IVD development.
Explore how to:
- Build and validate your workflow on the Seven Bridges analysis platform
- Deploy your IVD’ s analysis pipeline to Velsera's Platform
- Run your clinical workflow with Velsera